New Publication: Journal of Physical Chemistry Letters 2021
Biophysical Chemistry, NMR Structural Biology, and Molecular Bioengineering Lab
Prof. Myriam CottenThe Cotten lab has published an article in the Journal of Physical Chemistry (JPC) Letters, titled “Coordination of Redox Ions within a Membrane-Binding Peptide: A Tale of Aromatic Rings.” This excellent journal publishes short reports featuring “significant scientific advance and/or physical insight”. In the case of the Cotten lab, the study employs solid-state nuclear magnetic resonance and quantum calculations to identify stabilizing molecular interactions important to the anti-infective function of host defense peptides. Graduate student Mary Rooney contributed to this work. This research has relevance to a broad range of peptides and provide principles that could be used for applications in areas such as drug-resistance pathogens, neurological diseases, and biomedical imaging.”
Abstract:
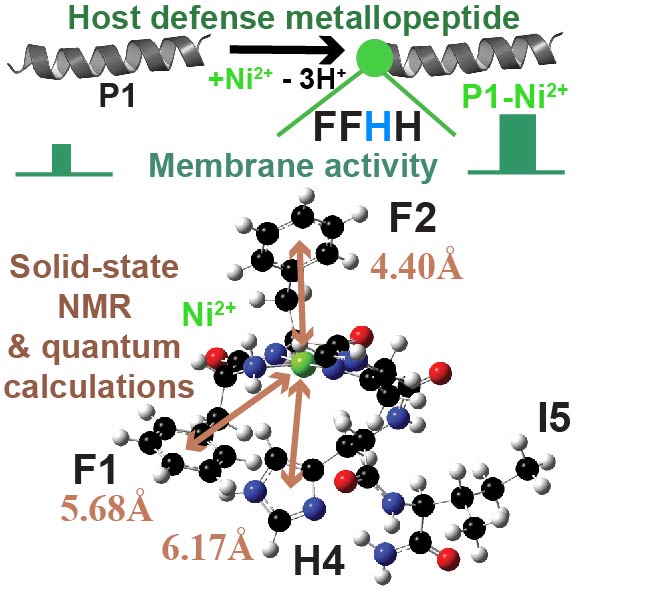
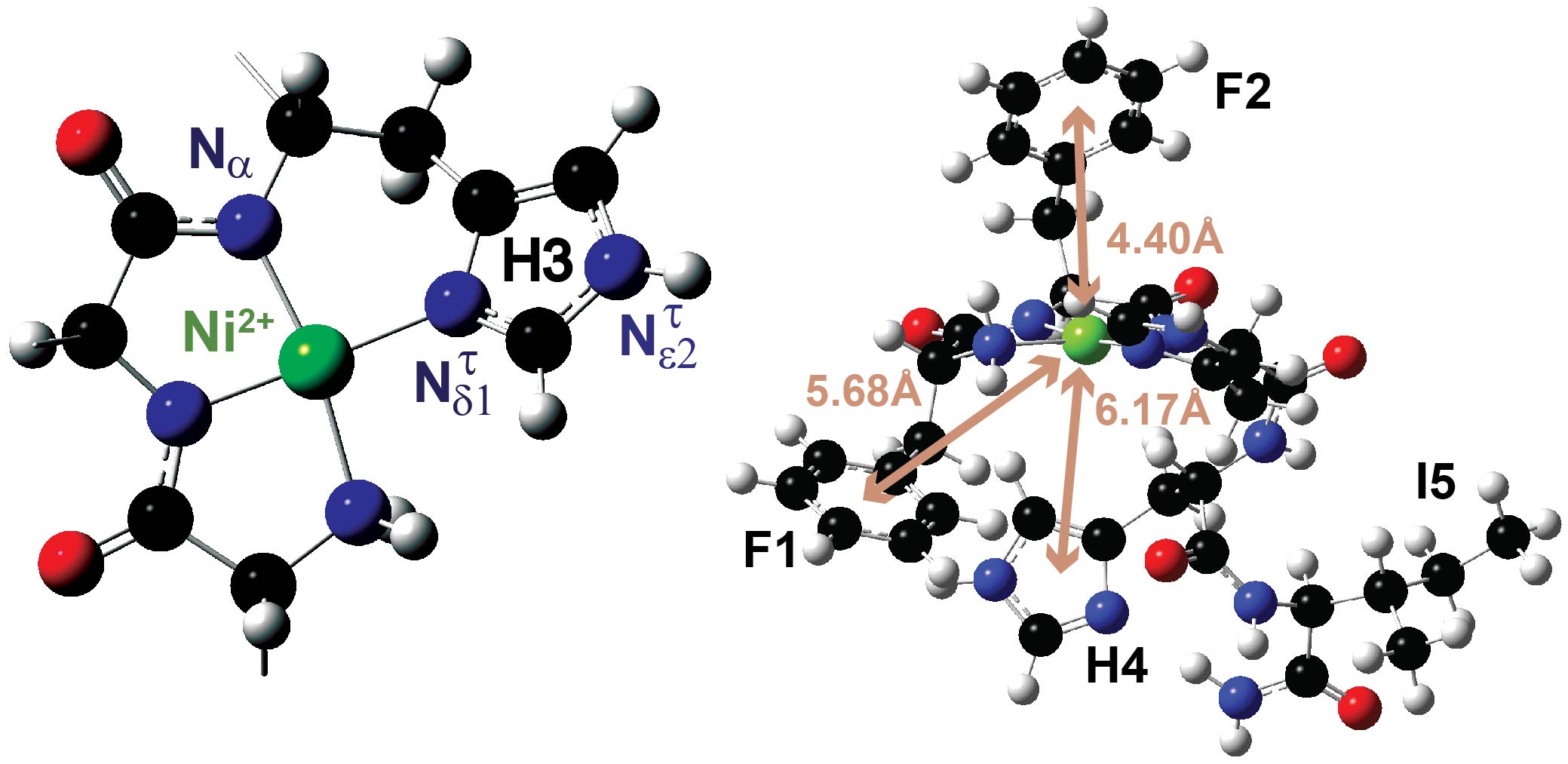
The amino-terminal-copper-and-nickel-binding (ATCUN) motif, a tripeptide sequence ending with a histidine, confers important functions to proteins and peptides. Few high-resolution studies have been performed on the ATCUN motifs of membrane-associated proteins and peptides, limiting our understanding of how they stabilize Cu2+/Ni2+ in membranes. Here, we leverage solid-state NMR to investigate metal-binding to piscidin-1 (P1), a host-defense peptide featuring F1F2H3 as its ATCUN motif. Bound to redox ions, P1 chemically and physically damages pathogenic cell membranes. We design 13C/15N correlation experiments to detect and assign the deprotonated nitrogens produced and/or shifted by Ni2+-binding. Occupying multiple chemical states in P1-apo, H3 and the neighboring H4 respond to metalation by populating only the τ-tautomer. H3, as a proximal histidine, directly coordinates the metal, compared to the distal H4. Density functional theory calculations reflect this noncanonical arrangement and point toward cation−π interactions between the F1/F2/H4 aromatic rings and metal. These structural findings, which are relevant to other ATCUN-containing membrane peptides, could help design new therapeutics and materials for use in the areas of drug-resistant bacteria, neurological disorders, and biomedical imaging.
Citation:
Riqiang Fu*, Mary T. Rooney, Rongfu Zhang, and Myriam L. Cotten*Publication Date: May 3, 2021
2021 American Chemical Society
Links:
Biophysical Chemistry, NMR Structural Biology, and Molecular Bioengineering Lab
 Skip to main content
Skip to main content