At the symposium: Two lasers and a focus on climate change
The center of our solar system is not located in Room 1243 at William & Mary’s Integrated Science Center, but if you’re a molecule of brown carbon, it may as well be.
The chemistry lab has been designed to mimic the way molecules interact with the sun. It is equipped with two lasers that emit wavelengths matching the sun’s UV frequencies. The lasers are fired at molecules pulsed in to a vacuum chamber.
When the lasers strike a molecule, it breaks apart and the fragments enter an electric field created by charged metal plates. The field directs the fragments down to a detector, which captures an image of what the products look like when the molecule interacts with sunlight.
 For Kenneth Blackshaw, a first-year master's degree candidate in the chemistry department at William & Mary, the lab has offered him the opportunity to study the elements that populate Earth’s atmosphere. He has spent the better part of the past year zapping brown carbon molecules to see how the particles, primarily created by fossil fuel emissions, break down with sunlight. The breakdown process is known as “dissociation.”
For Kenneth Blackshaw, a first-year master's degree candidate in the chemistry department at William & Mary, the lab has offered him the opportunity to study the elements that populate Earth’s atmosphere. He has spent the better part of the past year zapping brown carbon molecules to see how the particles, primarily created by fossil fuel emissions, break down with sunlight. The breakdown process is known as “dissociation.”
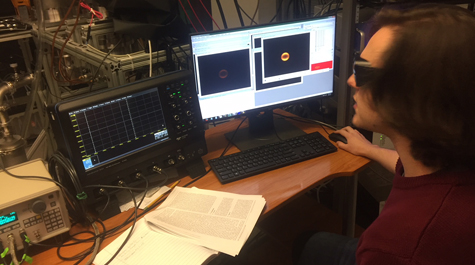
“This is pretty much where I live. Right here on this seat,” Blackshaw said, sitting in front of a series of imaging monitors. One clear image of a brown carbon molecule in the dissociation process requires roughly 30,000 shots, he explained.
The long hours have finally paid off. Blackshaw’s research into brown carbon has earned him the Graduate Studies Advisory Board’s prestigious “Excellence in Scholarship” award. He will receive the award on March 17 at William & Mary’s 17th annual Graduate Research Symposium. Blackshaw will present his research at 3:30 p.m. on March 16 in Chesapeake ABC.
“It’s reassured my faith in the research that we do here,” Blackshaw said of the award. “We’re trying to emulate what the sun does and the way radiation breaks apart molecules. With our research, we can determine how much light is required, how long this dissociation process takes and what actually is formed from the process.”
Blackshaw’s eventual goal is to provide the research to climatologists, who will use the data to predict changes in the Earth’s climate. He says his research would not be possible without his advisor, Nathan Kidwell, assistant professor of chemistry, as well as a team of undergraduate students.
“We’re working to get a better understanding of the physical properties of these molecules,” Blackshaw said. “That will eventually help us determine better climate models.”
Until relatively recently, brown carbons were not taken into account along with black carbon measurements, Blackshaw said. Black carbons have been extensively studied and are a known contributor of greenhouse gases, which have been linked to global climate change. Like black carbon, brown carbon molecules primarily result from air pollution, specifically burning fossil fuels. They are also formed from natural events like forest fires. While the two carbon molecules are similar, they differ in how they react to sunlight.
“Brown carbons were assumed to have the same absorption properties as black carbons until people took a closer look,” Blackshaw said. “What they found was that black carbons absorb almost all the radiation from the sun, but brown carbons don’t absorb all the radiation. They absorb more specific wavelengths more strongly.”
What this means, Blackshaw explained, is current climate models don’t account for brown carbon and the way it breaks down in the atmosphere. For example, brown carbon absorbs a high level of radiation from UV rays. The energy from that absorption is enough to break its molecular bonds. When the molecules break apart, they release new molecular fragments that are highly reactive and can contribute to the formation of ozone, a known tropospheric pollutant.
Brown carbon also absorbs visible radiation, which has an overall warming effect on the atmosphere. Blackshaw said brown carbon is classified as a greenhouse gas due to the high level of visible radiation it absorbs. He added that even the slightest change in the atmosphere can have a huge ripple effect – and it’s not always easy to predict whether the result will be positive or negative.
“I’ve been doing atmospheric research for a couple of years now and a big thing I’ve learned is that the atmosphere is a balance,” Blackshaw said. “There’s a lot of different molecules floating around and you can’t say ‘This thing is definitely causing destruction,’ because it might be doing some good, too. It’s a balancing act.”
One thing is clear to Blackshaw, when the atmosphere is off balance, a changing climate is the result. He says it’s a challenge to get the general public to accept the atmospheric science at the root of global climate change. And often, the effect grabs public attention while the cause takes a backseat.
“Strong visuals like a melting glacier or a polar bear on a thin sheet of ice, those are important because they are symptoms that we can see,” Blackshaw said. “When it comes to the actual cause of global climate change, it’s just as important, but it’s a little bit harder to understand.”
One way to make atmospheric science easier to understand is to make research more accessible, Blackshaw says. That was his goal when he submitted an abstract for consideration for the Graduate Research Symposium award. His victory, he said, was a validation.
“It means that people are interested in the work that we do here and are impressed by the research that’s going on,” Blackshaw said. “That’s really inspiring to me as a budding atmospheric chemist.”
 Skip to main content
Skip to main content